 
There are no disease modifying therapies approved for GA to date. It is estimated that there are over one million individuals with GA in the United States according to published data. Geographic atrophy is a highly prevalent disease with significant unmet medical need. 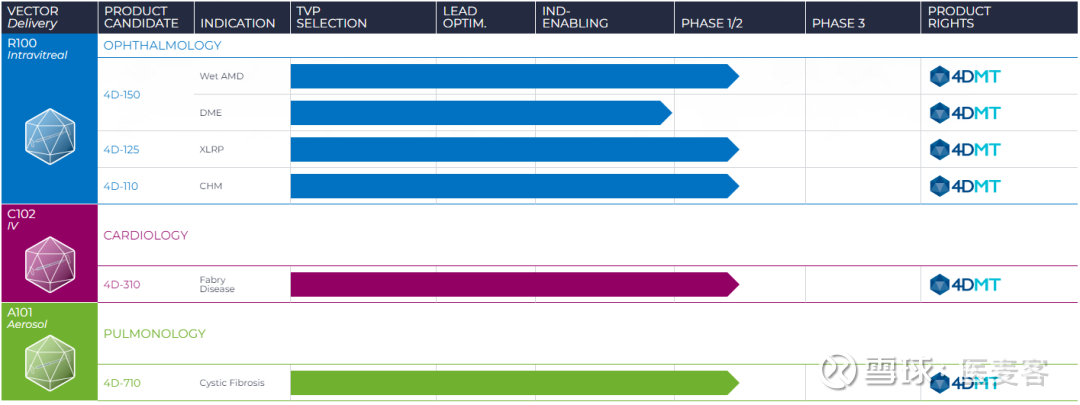
This Phase 2 stage of the trial was initiated in January 2023.Įxpanded Portfolio with Preclinical Product Candidate 4D-175 for Geographic Atrophy (GA): Patients will be randomized in masked fashion to receive a single intravitreal injection at one of two dose levels of 4D-150 (3E10 and 1E10 vg/eye) or aflibercept in a 2:2:1 ratio (n=50 patients). The Phase 2 stage of the Phase 1/2 PRISM clinical trial of 4D-150 in patients with wet AMD consists of three treatment groups. Initiated Randomized Phase 2 of PRISM Clinical Trial of Intravitreal 4D-150 in Patients with Wet AMD: 
We expect to initiate enrollment in the third quarter of 2023. The doses to be evaluated in DME are expected to be similar to those used in the 4D-150 wet AMD clinical trial. The Phase 2 SPECTRA clinical trial design consists of a Dose Confirmation stage followed by a masked Dose Expansion stage in which patients will be randomized to receive a single intravitreal injection at one of two dose levels of 4D-150 or aflibercept in a 1:1:1 ratio (n=54 patients). Ophthalmology Product Candidate PortfolioĤD-150 for the Intravitreal Treatment of Patients with Wet Age-Related Macular Degeneration (wet AMD) and Patients with Diabetic Macular Edema (DME)įiled IND Application for Phase 2 SPECTRA Clinical Trial with Intravitreal 4D-150 in Patients with DME:ĤDMT filed an Investigational New Drug (IND) Application for 4D-150 in patients with DME in December 2022, following pre-IND correspondence and alignment with the FDA. We are pleased to announce the strategic decision to expand our large market indication portfolios in ophthalmology and pulmonology using vectors that have already been used in product candidates dosed in humans.” 
“Given the promising early clinical trial data with 4D-150 in wet AMD and 4D-710 in cystic fibrosis lung disease that we announced in Q4 2022, plus cardiac endpoint clinical trial data reported today with 4D-310, we believe the broad potential of our platform is evident. “These updates highlight 4DMT’s diversified and growing product pipeline that is driven by our underlying directed evolution vector platform and our robust product design and development engine for genetic medicines,” said David Kirn, M.D., Co-founder and Chief Executive Officer of 4DMT. (Nasdaq: FDMT), a clinical-stage biotherapeutics company harnessing the power of directed evolution for targeted genetic medicines, announced product pipeline portfolio updates and preclinical product candidate additions for its large market ophthalmology and pulmonology programs, as well as clinical data and program updates for its 4D-310 Fabry disease program. 09, 2023 (GLOBE NEWSWIRE) - 4D Molecular Therapeutics, Inc. Cash Guidance Unchanged and Sufficient to Fund Operations into H1 2025ĮMERYVILLE, Calif., Jan.Reports Program Updates and Interim Clinical Data from Phase 1/2 Fabry Disease Cardiomyopathy Trials Full Clinical Data to be Reported at WORLDSymposium in February 2023.Expands Large Market Pulmonology Portfolio Following Positive Clinical Data in Cystic Fibrosis to Include Alpha-1 Antitrypsin Deficiency Preclinical Program.Expands Large Market Ophthalmology Portfolio Following Positive Clinical Data in wet Age-Related Macular Degeneration to Include Diabetic Macular Edema Clinical Program and Geographic Atrophy Preclinical Program. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
